How to use CellTank-34
5 easy steps to single-use Continuous-Perfusion in CellTank-34.
- Remove SUB from plastic bag
- Connect sterile media bag and harvest bag to the SUB
- Pump media into the SUB
- Connect and calibrate the pre-installed classical SUS
- Inoculate suspension or adherent mammalian cells and start cultivation
----------------------------------------------------------------------------------------------------------------------------------
The following is recommendations only.
- First SOP dated 4th of January 2012, version 3.1
- Second SOP dated june 2018, version 4.0
We suggest you develop your own Standard-Operation-Procedure (SOP).
----------------------------------------------------------------------------------------------------------------------------------
Install CellTank-34 on the MST and inside the HSF-2266-E mounted on the MST. DO NOT start agitation before media is added covering at least 50% of planned total volume. (centrifugal pump bearing media lubricated)
Preparation with latest Single-Use-Sensor's (SUS - 2018):Pre-installed Single-Use-Sensor's in PG 13,5x120 ports in CellTank-34:
- DO VisiWell in port #10 only
- pH OneFerm in port #11 only
- C-CIT Glucose in port #12 only
- PICO bio-mass SUS in port #13 only
Appropriate cabling from your PCS and pre-amplifiers from C-CIT and Aber.
Preparation with Single-Use-Sensor's (SUS) in CellTank-34:
- Insure your have the appropriate cabling to the pre-installed and pre-calibrated Hamilton classical signal pH SUS.
- Install the Hamilton classical signal VisiFerm PG 13,5x120 sensor body (not part of the CellTank kit) without the;1. Cap, 2.the tiny O-ring. 3. the large O-ring under the PG 13,5 thread into the VisiWell. Insure your have the appropriate cabling between the Hamilton RUS and you PCS. Install and calibrate appropriate the sensor according to the calibration chart found in the package.
- Install the Aber PICO pre-amplifier (not part of the CellTank kit) on top of the bio mass connector and cabling to the instrumentation (not part of the CellTank kit) according to spec from Aber.
- Install the C-CIT blue pre-amplifier to the Glucose SUS and establish the Wi-Fi connection.
Pre-cautions:
- DO NOT heat up the SUB until after media is added!
- Insure correct location of the SUB on the Magnetic-Stirrer-Table (MST)
- DO NOT dry-run the integrated impeller with the MST!
- DO NOT stop the re-circulation pump at any time as this will stop the media flow through the matrix containing the cells
- The product is designed for single-use only and will be damaged during treatment with ethanol or temperature above 50°C.
Media considerations
Serum containing media will create more foam compared to chemical defined (CD) media.
Use of external foam collecting vessel size is depending on the serum content. 5 - 10% requires typically an external foam trap vessel for protection of the exhaust gas sterile filter. 2% serum containing media operates typically without external foam collecting vessel. Foam generation is heavily depending on the sparging gas volume and typically seen above 3 l/h.
Prep before start-up
- Calibrate the Hamilton DO sensor with air in the reservoir
- Connect appropriate sized (>25 litre) media bag to port #2 and harvest bag to port #3
- Ad 1,000 ml media to the SUB under sterile conditions. The CellCore body volume is app 280 ml. 1,000 + 280 = app 1,280 ml level in vessel. Insure the sensors are fully covered with media. Remember the inoculation volume will ad to the reservoir level!
- Install the CellTank accurately at the centre on the clockwise rotating stirrer table
- Add 5-10 ml water to the non-invasive sensor port #8
- Mount the thermocouple in the non-invasive port #8
- Mount appropriate heating blanket around the vessel
- Install the SS rod in the rod well on the cover and mount the sky-support
- Expand the WC tube from port #16 vertically. Insure the hose clamp is locked. Insure the OD25 mm sterile filter is not blocked, mount the filter on sky-support.
- Connect sparging gas supply to port 7 OD50 mm sterile filter, install optionally filter heater and secure the filter mechanically vertical
- Connect head space flushing gas supply to port #6 OD50 mm sterile filter and secure the filter mechanically vertical
- Initiate the MST and increase the rpm until you see the rotameter flotation device lifts inside the rotameter. Watch the CellCore is de-aired as gas and bubbles are exhausted through the tiny hole in the central rod.
- Obtain stable condition at 0.8 litre/min flow (8 cm/min flux) on the re-circulation pump typically ranging 250-260 rpm.
- Obtain stable conditions such as temperature
- Calibrate the Hamilton SUS pH sensor according to the calibration chart found in the package.
- Test three times for traction loss rpm in order to understand the MST potential. Increase with 5 rpm / second until the level is reached where the flotation device in the rotameter drops down and the SUB shakes. Make a note of the average traction loss rpm and keep 10% rpm distance below at any time.
- Return to 0 rpm and increase to 0.8 litre/min flow typically 250-260 rpm, insure the media is flowing over the rotameter.
- Open the hose clamp on the WC tube and insure the filter is not blocked.
- Inoculate through port #1 or port #2 and start the cultivation in batch mode
Some CellTank´s are pre-equipped with a 500 ml foam collection vessel which may be installed hanging from the sky-support with filter heater on the exhaust sterile filter.
Gas control
- Sparging gas ranging 3-6 l/h mixed gas 10-100% O2 / 90-0% N2
- Head space flushing gas range 4-8 l/h air, head space gas pressure >0.5 mBar for controlled pCO2, partial pressure in the media ~5 kPa.
Liquid level control
On Process-Control-Systems with no balancer but calibrated peristaltic pumps:
- Use one pump to supplying fresh media (100% volume) to one of several inlet port. Harvest the product containing media (95% volume) from the bioreactor to suitable harvest storage through harvest port. Use one pump (10% volume) connected to liquid level port which then insures constant media level with return to harvest storage.
- Use the pre-installed level sensor to control a peristaltic pump on/off or rpm operation. Remember that with SUS there is a need for an artificial ground to one of the electrodes taken from the PCS cabinet ground.
Inoculation of cells
High density – for the new user!!
Insure the cells are suspended carefully at high density in 50-100 ml media. Cell numbers to be inoculated depends heavily on the cell line and range for CHO cells 0.1-2x10E+06 cells/ml/matrix.
You may choose one of several routes for inoculation:
- Into the reservoir through port #5 or 6
- Into the volume between the impeller and the matrix through port #2
The impeller operating for a 0.8 l/min will distribute uniformly the cells inside the matrix.
Low density – for the experienced user!
A shaker flask with 1,000 ml media is used to grow up the cell line from frozen stage in an incubator at 37°C. Exchange the cap with a cap integrating a silicone hose and weld onto a suitable hose on the CellTank. Pump in the media in 10 minutes and set temperature on the PCS to 37°C. Start the re-circulation pump in the CellTank when +800 ml is transferred for 1 l/m flow.
Separation of cells:
- Suspended cell line collection inside the matrix typically takes between 15 and 120 minutes. Hereafter the media is clear again and contains from 500 thousands down to thousands of cells/ml.
- Adherent cell lines separate by various collection phenomena inside the matrix and typically take between 5 and 50 minutes. Hereafter the media is again clear and contains from thousands down to only hundreds of cells/ml.
Cell trapping control
The build-in impeller insures by rotation a constant re-circulation of media through the matrix.
- Suspension cell lines stay trapped inside the matrix cavities when media is constantly re-circulated at at least 0.8 l/m - avoid rapid media flow changes.
- Adherent cells stays trapped inside the matrix independent of the impeller is rotating or not – well, after seeding!
In any case media flow is needed in order to avoid gradients and volume determined by the cell mass, dO2, glucose and lactate content among others. Do not reduce the flux to a level below the requirements of the cells.
preferred_SOP
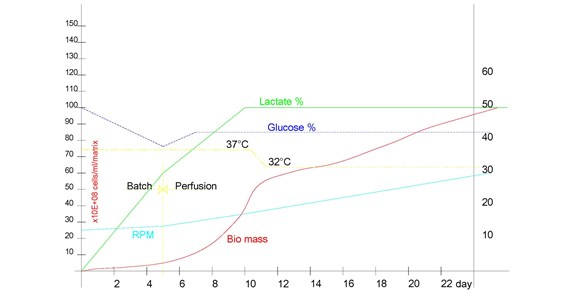
Figure – Cultivate in batch up to lactate / glucose decides perfusion start. Cultivate up to 50x10E+06 cells/ml/matrix and continue in perfusion. Reduce temperature with >5°C or more when lactate reaches 100% allowable and when glucose drops below acceptable levels.
Batch cultivation
Start cultivation after inoculation in batch mode at >0.8 l/m re-circulation flow for 3-5 days and grow up to ~50x10E+06/ml cells/ml/matrix. Remember that correction factor between suspension cell to pF/cm is different from adherent cell lines.
The increasing high amount of cells will produce plenty of CO2 and in order to reduce or reduce the addition of acid the head space must be flushed properly.
Suspension cell proliferation under perfusion mode operation
Start the perfusion when glucose / lactate drops / increases beyond your acceptance. And at ~50x10E+06/ml cells/ml/matrix reduce the SUB temperature from 37C to a suitable level such as 32-30°C or lower in order to reduce proliferation.
Flux decrease dynamically according to increased cell density and range between 8 - 20 cm/min. Media replacement start from scratch and increase to as much as 30 matrix volumes per day depending on cell mass and cell type.
Expect a needed increase in impeller rpm each day in order to overcome the increased back pressure as to the increase cell density. Such as 10% increased rpm per day and an increase from start at 260 rpm to 500 rpm or more over 14 days operation. Be aware of the particular brand MST capability in use.
Maximum operational cell density is determined by the MST traction capability and varies between 500 and 750 rpm which are depending on brand! It is VERY important NOT to reach the traction loss rpm as the process will stop! Check traction guide lines.
DO NOT target 1x10E+08 cells/ml/matrix (unless you have such experience) before you reduce the SUB temperature.
Cell mass control
There is several ways of controlling the maximum suitable bio mass in the matrix.
- Rapid increased impeller rpm (such as 50-100 rpm increase/second) will rip off/out high amount of suspension cells in the matrix to the reservoir. This high amount of cells in the reservoir is also known as “bleeding cells” from the SUB and may be taken out of the SUB through a suitable port. Cells in the reservoir will be trapped again after 15 and 120 minutes at suitable flux.
- Decrease the proliferation by altering the temperature to a lower level than 37°C on both adherent and suspension cell lines
- Decrease the proliferation by altering the glucose level on both adherent and suspension cell lines
Suspension cell perfusion cultivation
Max cell density and the preferred method of keeping the cell density in control is the equilibrium for the production stage.
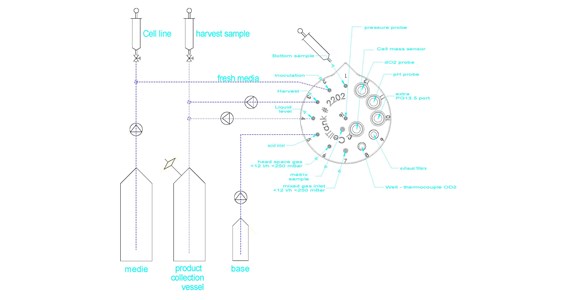
Suggestions to the actual set-up
layout-suspension-no-serum
Figure 4 – The illustrated set-up for CD media with four pumps controls the reservoir liquid level based on level sensed by conductivity via the level sensor.
Download here the above figure as a pdf file here.
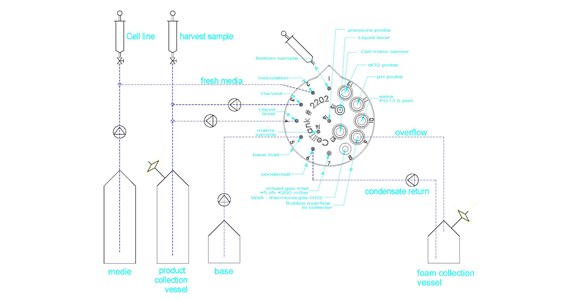
layout-suspension-serum
Figure 5 – The illustrated set-up for serum containing media with five pumps determines the reservoir level as an alternative to a balancer or level control by conductivity. Port 9 leads foam to foam collection vessel which also then isolated the sterile exhaust filter from foam. Condensate foam is as a liquid re-directed to the vessel.
Download here the above figure as a pdf file here.
Sampling
On three ports swabable valve with cover for sterile sampling are fitted.
Data collection
We suggest the following data is collected continuously. Further some suggested appropriate ranges for CHO cell lines:
- pH -
- dO2 –
- bio mass – 0-250 pF/cm (able to measure 700 pF/cm)
- Matrix sample, viable cell density
- Harvest sample, viable cell density
- Bottom sample, viable cell density
- Glucose – 10-20 mM
- Lactate – 5-50 mM
- Glutamine – 1.5-2-5 mM
- Glutamate – 0.5-1.5 mM
- Twice per day the re-circulation volume - 1-2.5 l/min
- Twice per day the WC – matrix pressure drop in mm Water Column – 100-500 mm
- Ammonia – 3-5 mM
- pCO2 – partial CO2 pressure 2-5 kPa in the reservoir controlled from head space
- Perfusion rate - reactor/volume – RV/day
PerfuseCell further recommend looking at the Application Notes